Acid Rain's Effect on Marble |
|||
|
Author(s): Jim Schwagle, Kevin Bryan |
Growth & Development
Experiment |
||
|
Research Question: What effect does acid rain have on marble?
|
|
||
|
Standards addressed: High School Chemistry
|
|||
|
Independent variable |
Dependent variables |
Controls |
Series |
|
Time |
Change in mass of marble samples |
Marble sample with application of distilled water |
pH 4, 5, 6 and distilled water |
|
Materials |
Procedures |
||
|
|
||
|
|
|||
|
|
Blank Data table |
||
|
|
Data table with starting masses, mass each week and % mass lost each week. |
||
|
Click map for larger image at the source site
|
pH values for rain across the country. |
||
|
|
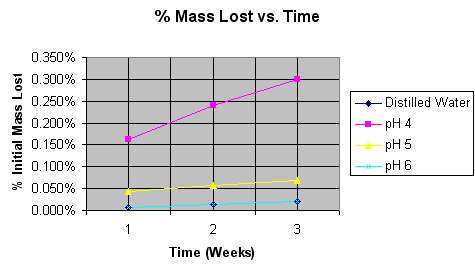
This graph depicts the percentage of starting mass lost over time. Although distilled water seems to be missing, it is in fact underneath, and virtually identical to pH 6. |
||
|
|
This statue has been slowly destroyed by acid rain so that now the faces are barely visible. |
||
|
References & Links: National Atmospheric Deposition Program Deposition Maps (Excellent maps of deposition of many different ions nationwide)
|
|||